Please note: a valid prescription is required for all prescription medication.
Buy Fiasp FlexTouch online with a valid prescription and compare current listed pricing, pen presentation details, and key safety basics before ordering. You can check the selected form, U-100 strength, quantity, and handling needs while matching the listing to your clinician’s directions.
If you are comparing Fiasp FlexTouch US delivery from Canada, review the product selection, cold-chain considerations, and any checkout details before you proceed. Keep your prescriber information available in case order details need confirmation.
Fiasp FlexTouch Price and Available Options
The Fiasp FlexTouch price shown on the listing should be compared with the selected pen presentation, quantity, and total units supplied. Current listed pricing can change when a different quantity is chosen or when another Fiasp format is listed separately.
Look beyond the product name before adding the item to your cart. Fiasp may be available as a prefilled FlexTouch pen, cartridges, or vials, and those formats are not interchangeable for every patient. A pen listing is intended for people prescribed the FlexTouch device, while cartridges require a compatible reusable pen system and vials require a syringe or pump plan when appropriate.
Because this is U-100 insulin, the concentration is 100 units per mL. A common 3 mL pen contains 300 units total, but that total content is not a single dose or a suggested amount to inject. Your selected quantity should reflect the number of pens or packs listed, not a change to your prescribed units.
If you are comparing Fiasp FlexTouch cost without insurance, check the cash-pay total at checkout and confirm whether supplies such as pen needles are listed separately. Coverage status, selected quantity, and product format can all affect the final amount you review before placing an order.
Quick tip: Match the product form first, then compare quantity and total units.
How to Buy Fiasp FlexTouch Online
To buy Fiasp FlexTouch online, start by confirming that your prescription names the prefilled pen rather than another Fiasp presentation. The product title, concentration, and quantity should align with the directions your clinician provided.
- Presentation check: confirm FlexTouch pen, not cartridge or vial.
- Strength check: match U-100, 100 units per mL.
- Quantity check: compare pens or pack contents before checkout.
- Prescriber details: keep clinic contact information available if needed.
- Supply check: confirm whether compatible pen needles are separate.
Prescription details may be verified with your prescriber when needed. Supporting documents may be requested for some orders, especially when product details or directions need clarification.
The order path is easier when the selected product matches your current insulin plan. If you use pen needles, the Insulin Pen Needles resource can help you review needle basics before selecting supplies.
Pen Presentation and Strength Details
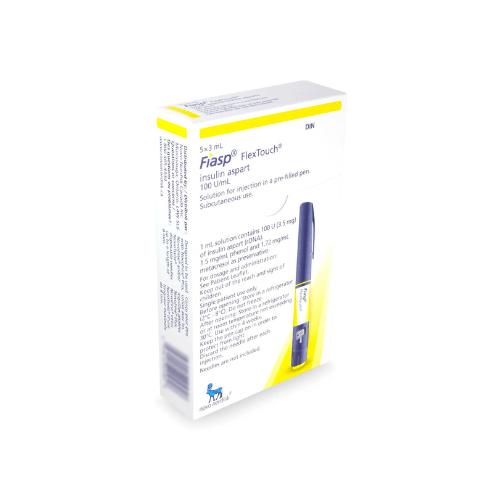
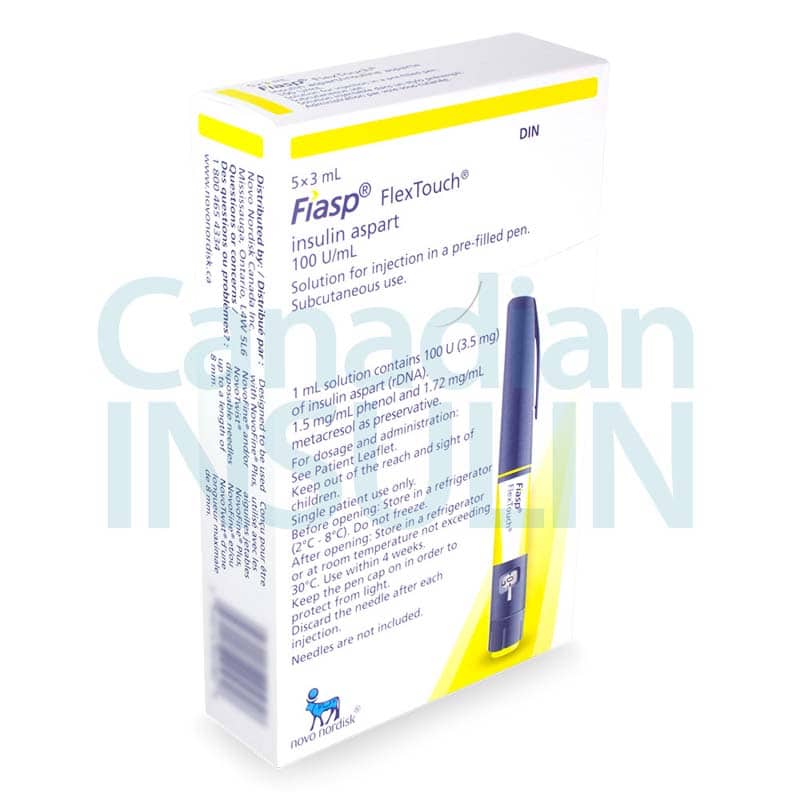
The Fiasp FlexTouch insulin pen is a prefilled device for subcutaneous injection under the skin. It contains insulin aspart, a rapid-acting insulin analog (modified insulin designed to work quickly), used around meals as directed by a clinician.
| Product detail | What to check |
|---|---|
| Active ingredient | Insulin aspart |
| Insulin class | Rapid-acting prandial insulin for mealtime use |
| Concentration | U-100, meaning 100 units per mL |
| Presentation | Prefilled FlexTouch pen, commonly 3 mL |
| Total contents | A 3 mL pen contains 300 units total |
| Dose selection | Match prescribed units and do not estimate from volume |
| Needle use | Use a new compatible needle for each injection |
The Fiasp FlexTouch U-100 insulin pen is designed for dose selection in units. Audible clicks during dialing may help some users confirm movement of the dose selector, but the dose window should still be checked visually when possible.
Your Fiasp FlexTouch dose should match your prescribed insulin plan. Do not adjust Fiasp FlexTouch dosing based on product contents, remaining insulin in the pen, or another person’s regimen. If the device, dose window, or insulin appearance seems wrong, pause and contact a healthcare professional.
What This Rapid-Acting Insulin Is Used For
Fiasp is used to improve blood glucose control in people with diabetes who need rapid-acting insulin. It is often used around meals, because it helps move glucose from the bloodstream into cells after food intake.
This medicine is not a basal insulin. Basal insulin provides background coverage over a longer period, while a mealtime insulin works over a shorter window. Many people with type 1 diabetes use both types, and some people with type 2 diabetes may use rapid-acting insulin as part of a broader plan.
A Fiasp rapid acting insulin pen should be used only as directed. Timing, carbohydrate intake, activity level, illness, and other medicines can change insulin needs, so the product listing should never be used to choose a new regimen.
Storage, Handling, and Travel Basics
Insulin is temperature-sensitive, so storage matters before and after delivery. Unopened pens are generally kept refrigerated at 2°C to 8°C, or 36°F to 46°F. Do not freeze insulin, and do not use a pen that has been frozen.
After first use, Fiasp FlexTouch is usually kept at room temperature under label storage limits and discarded after the allowed in-use period, often 28 days. Follow the product carton and patient information for the exact storage instructions supplied with your order.
Because insulin can be affected by heat and freezing, checkout logistics may use express, cold-chain shipping rather than standard handling. Inspect the package when it arrives, refrigerate unopened pens as directed, and avoid leaving insulin in a hot car, checked luggage, direct sunlight, or near freezer packs.
For a focused review of temperature ranges and common storage mistakes, keep Insulin Storage Temperature available with your diabetes supplies. Travel planning should include a backup pen, extra needles, glucose monitoring supplies, and the original labeled packaging.
Safety Checks Before Buying
Fiasp is contraindicated (should not be used) during episodes of hypoglycemia (low blood sugar) or in people with hypersensitivity to insulin aspart or any ingredient in the product. Check the label and your clinical history before selecting the listing.
The most important risk with any insulin is hypoglycemia. Symptoms may include shakiness, sweating, fast heartbeat, hunger, confusion, weakness, or blurred vision. Severe low blood sugar can cause seizures, loss of consciousness, or require emergency treatment.
Other possible adverse effects include injection site reactions, itching, rash, weight gain, swelling, and changes in skin tissue at injection sites. Rotating injection sites within the recommended area can reduce the risk of lipodystrophy or localized cutaneous amyloidosis, which are changes in the skin or fatty tissue that can affect insulin absorption.
- Do not share pens: infection can spread even with a new needle.
- Check appearance: do not use cloudy, colored, or particle-filled insulin.
- Watch potassium risk: insulin can lower blood potassium in some patients.
- Seek urgent help: severe allergy, breathing trouble, or fainting needs care.
Why it matters: Low blood sugar can impair driving, work, and daily decisions.
The Low Blood Sugar resource can support symptom recognition, but your clinician’s plan should guide treatment steps.
Interactions, Monitoring, and Dose Questions
Several medicines can change how insulin affects blood glucose. Corticosteroids, diuretics, thyroid medicines, some psychiatric medicines, and decongestants may raise glucose levels in some people. Other medicines can increase the glucose-lowering effect and raise the risk of hypoglycemia.
Beta blockers and some blood pressure medicines may also mask warning signs such as a fast heartbeat. Alcohol, skipped meals, vomiting, unusual exercise, or changes in carbohydrate intake can make glucose harder to predict.
Tell your healthcare professional about all prescription drugs, over-the-counter medicines, supplements, and alcohol use before using a Fiasp insulin aspart pen. This is especially important if you use thiazolidinediones, because combining those medicines with insulin can increase fluid retention and may worsen heart failure in susceptible patients.
Monitoring usually includes blood glucose checks, continuous glucose monitor readings when used, and awareness of patterns around meals. If readings are repeatedly high, ketones are present, or lows occur often, your clinician may need to reassess the plan. Do not change the prescribed dose or timing based only on an online product description.
Compare With Related Insulin Options
Fiasp FlexTouch, cartridges, and vials may contain the same brand of insulin, but they differ in device workflow. If your prescription names a cartridge or vial rather than a pen, compare Fiasp Cartridges or Fiasp Vials instead of selecting the FlexTouch format.
Fiasp is not the same product listing as NovoLog or NovoRapid, even though these products are related to insulin aspart. Fiasp includes formulation ingredients intended to support faster early absorption. Differences in timing, device, labeling, and availability mean switching should be handled through a clinician, not by product comparison alone.
If your prescription mentions another rapid-acting option, the Rapid Acting Insulin category can help you compare product forms before choosing a listing. A Fiasp FlexTouch alternative may use a different active ingredient, device, concentration, or meal timing instruction.
Authoritative Sources
The clinical and device details above should be checked against official product information. Use the following resources for label-aligned information, not personal dosing decisions.
- Official prescribing and administration details are available from Fiasp Administration Options.
- Manufacturer patient instructions for the pen are available from FlexTouch Pen and Dosing Options.
This content is for informational purposes only and is not a substitute for professional medical advice.
Express Shipping - from $25.00
Shipping with this method takes 3-5 days
Prices:
- Dry-Packed Products $25.00
- Cold-Packed Products $35.00
Standard Shipping - $15.00
Shipping with this method takes 5-10 days
Prices:
- Dry-Packed Products $15.00
- Not available for Cold-Packed products
What type of insulin is Fiasp FlexTouch?
Fiasp FlexTouch is a prefilled insulin pen containing insulin aspart, a rapid-acting insulin analog. It is generally used around meals to help manage blood glucose after food intake. The pen format is different from cartridges and vials, so the selected product should match the prescribed presentation. Fiasp is U-100 insulin, meaning it contains 100 units per mL.
Is Fiasp the same as NovoLog?
Fiasp and NovoLog are related because both involve insulin aspart, but they are not the same product. Fiasp has formulation ingredients intended to support faster early absorption, and its label, timing instructions, and device options may differ. A person should not switch between rapid-acting insulin products based only on name similarity. Any change should be reviewed with the prescribing clinician.
What are the main safety concerns with Fiasp FlexTouch?
The main safety concern is hypoglycemia, or low blood sugar. Symptoms can include shakiness, sweating, hunger, confusion, weakness, or blurred vision. Serious reactions can include severe allergy, fainting, breathing problems, or very low potassium in susceptible patients. Pens should never be shared, even if the needle is changed, because bloodborne infection can spread through shared injection devices.
What should I ask my clinician before using Fiasp FlexTouch?
Ask how the pen fits into your current diabetes plan, including meal timing, correction instructions, glucose monitoring, and what to do during illness or missed meals. It is also useful to ask how exercise, alcohol, other medicines, or steroid use may affect readings. If you have frequent lows, persistent highs, or trouble using the device, those points should be discussed before continuing the same routine.
How should Fiasp FlexTouch be stored?
Unopened Fiasp FlexTouch pens are generally refrigerated and protected from freezing. Once a pen is in use, it is usually kept at room temperature within label limits and discarded after the allowed in-use period, often 28 days. Keep insulin away from direct heat, sunlight, and freezer packs. Always follow the package insert and carton instructions supplied with the product.
Is the Fiasp FlexTouch pen being discontinued?
Availability can change by country, manufacturer supply, and pharmacy inventory, so discontinuation questions should be checked against official manufacturer or pharmacy information. If the pen format is unavailable, alternatives may include another Fiasp presentation or a different rapid-acting insulin. These options are not automatically interchangeable, because devices, timing instructions, and prescriptions may differ.
Rewards Program
Earn points on birthdays, product orders, reviews, friend referrals, and more! Enjoy your medication at unparalleled discounts while reaping rewards for every step you take with us.
You can read more about rewards here.
POINT VALUE
How to earn points
- 1Create an account and start earning.
- 2Earn points every time you shop or perform certain actions.
- 3Redeem points for exclusive discounts.
You Might Also Like
Related Articles
Non Hormonal Contraception: Options, Risks, and Fit
Non hormonal contraception means birth control that prevents pregnancy without using estrogen or progestin. Common options include the copper IUD, condoms, diaphragms, spermicides or contraceptive gels, fertility awareness-based methods, withdrawal,…
Weight Loss With Saxenda: Expectations, Risks, and Next Steps
Weight loss with Saxenda is usually gradual, not dramatic at the start. Saxenda is the brand name for liraglutide, a GLP-1 receptor agonist, which is a medicine class that can…
Semaglutide Weight Loss Medication: Safety, Options, and Expectations
A semaglutide weight loss medication is a GLP-1 receptor agonist (a hormone-mimicking drug that can reduce appetite) used in some settings to support chronic weight management. It changes hunger and…
What Are Sugar Alcohols? Sweeteners, Side Effects, and Facts
If you are asking what are sugar alcohols, the short answer is this: they are sweeteners called polyols that show up in many sugar-free or reduced-sugar foods. They are carbohydrates,…