Please note: a valid prescription is required for all prescription medication.
Lantus Vial is a prescription long-acting insulin used to help control blood sugar in diabetes. This product page helps patients compare how to buy or order it through a compliant process, with the main prescription, handling, and safety points laid out first. Some patients explore US delivery from Canada when local options, eligibility, and jurisdiction allow.
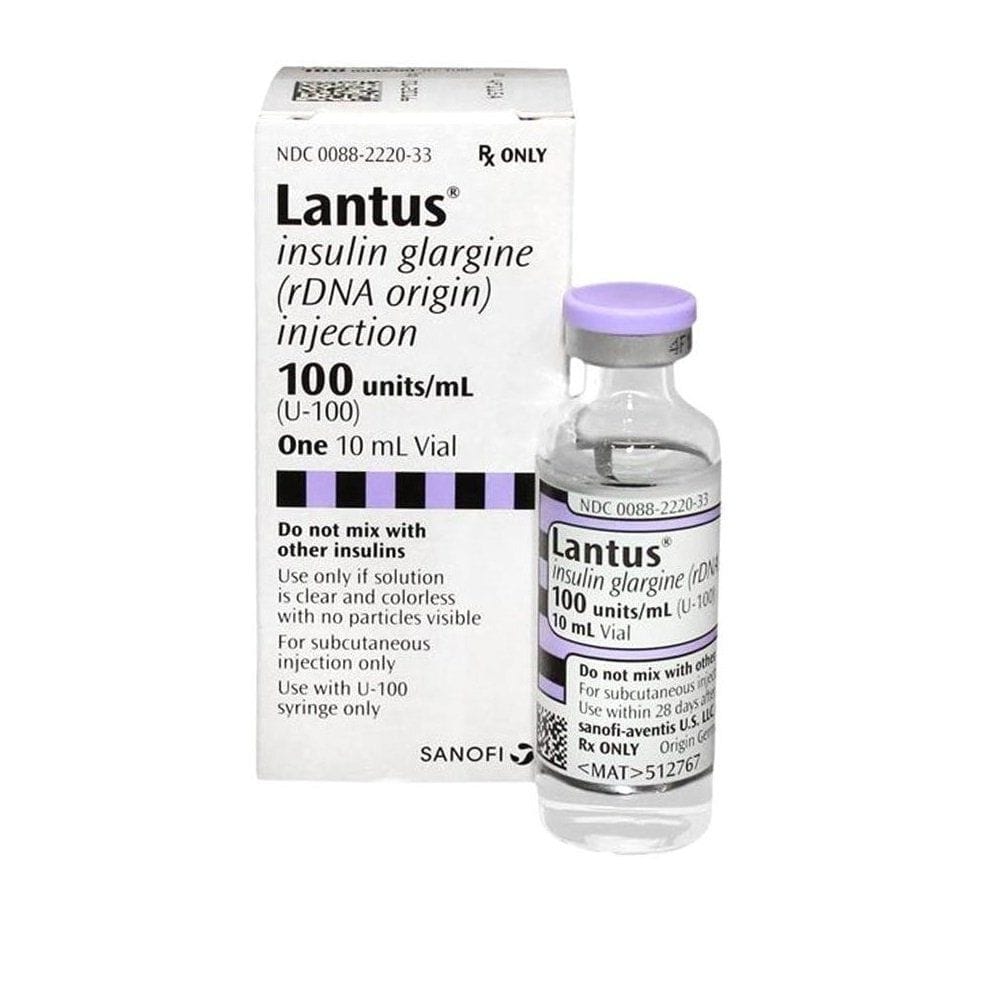
This listing is the 10 mL multidose vial form of insulin glargine U-100, not a pen device. Key questions usually involve who it may suit, how many units the vial contains, how long an opened vial lasts, and what precautions matter before use.
How to Buy Lantus Vial and What to Know First
This medicine is basal insulin (background insulin). It is used under the skin once daily to help steady glucose between meals and overnight. This listing refers to the multidose vial presentation rather than a pen, which matters because the prescription and supplies need to match the form being dispensed.
Prescription details may be checked with the prescriber when needed. Before pursuing a purchase, review whether the prescription names the vial, the daily units, and any companion supplies such as U-100 syringes. The main safety points to understand up front are low blood sugar risk, correct measuring technique, and storage that keeps the product within labeled conditions.
If a person has used only pens before, moving to a vial may require a practical review of dose measurement and site rotation. That is one reason this page focuses on access basics, handling, and safety first. For broader condition context, the Type 1 Vs Type 2 guide explains why insulin plans can differ across diagnoses.
Who It’s For and Access Requirements
Lantus Vial may be used by adults with diabetes who need a long-acting insulin, and it may also be prescribed for some pediatric patients when a clinician considers it appropriate. It is commonly part of type 1 diabetes care and may also be used in type 2 diabetes when oral medicines, lifestyle measures, or other injectables do not provide enough glucose control on their own.
For many people with Type 2 Diabetes, basal insulin is added when fasting blood sugar remains above target despite other treatment steps. In type 1 diabetes, long-acting insulin is usually part of a broader insulin plan rather than a stand-alone treatment. The right fit depends on diagnosis, current glucose patterns, prior insulin exposure, and the ability to use a vial and syringe accurately.
Access usually depends on a valid prescription that matches the vial presentation, strength, and dosing directions. Past severe low blood sugar, reduced vision, tremor, or limited hand dexterity can affect whether a vial and syringe is practical, so device choice may need review before dispensing.
Dosage and Usage
This insulin glargine product is generally taken once daily at the same time each day, but the exact number of units is individualized by the prescribing clinician. It is given by subcutaneous (under the skin) injection using a syringe drawn from the vial.
Common injection areas include the abdomen, thigh, or upper arm. Rotate sites within the same general area to reduce skin problems such as thickening, hardening, or pitting. Do not mix or dilute this insulin unless the official label specifically instructs otherwise, and never share needles or syringes with another person.
Because it acts across the day, missed doses, accidental double doses, or inconsistent timing can create unstable glucose patterns. Any catch-up instructions should come from the prescriber or official labeling rather than guesswork.
- Check the label first
- Inspect the liquid
- Use the prescribed syringe
- Measure the exact dose
- Rotate injection sites
- Track glucose as directed
Routine glucose checks help show whether the background insulin plan is matching daily needs. The Monitor Blood Sugar guide gives broader context on monitoring frequency and record keeping.
Why it matters: Taking a long-acting insulin at inconsistent times can make blood sugar patterns harder to interpret.
Strengths and Forms
Lantus Vial is the multidose 10 mL presentation of insulin glargine 100 units/mL. That concentration is often written as U-100, meaning each milliliter contains 100 units of insulin.
Questions about vial size are common because insulin is measured in units rather than milliliters. The concentration tells how many units each mL contains, while the fill volume tells the total units in the container.
| Attribute | Details | Why it matters |
|---|---|---|
| Concentration | 100 units/mL | Used to measure each dose |
| Fill volume | 10 mL | Defines vial size |
| Total insulin | 1,000 units | Answers the total-unit question |
| Route | Subcutaneous injection | Not for intravenous use |
| Container | Multidose vial | Drawn up with a syringe |
| Device | U-100 syringe if prescribed | Presentation and supplies must match |
That means a full vial contains 1,000 units in total. How long it lasts depends on the prescribed daily dose and the labeled in-use discard window. For example, a 20-unit daily regimen would use 50 days of insulin in theory, but an opened vial still follows the 28-day discard rule.
Pen devices may also be available for the same medicine, but they use different handling steps. The prescription must match the presentation because pens, pen needles, vials, and syringes are not automatically interchangeable.
Storage and Travel Basics
Unopened insulin glargine vials should be kept refrigerated and protected from freezing. If a vial has been frozen, it should not be used, even if it later looks normal.
After first use, the vial may usually be kept at room temperature within the labeled limits and should be discarded after 28 days, even if insulin remains. Marking the date first opened on the carton or vial label can make the in-use window easier to follow.
Keep the vial away from direct heat and light, and store it so the rubber stopper stays clean. During travel, insulin is best carried in an insulated case rather than packed loosely in a hot car or checked baggage.
Quick tip: Carry insulin near a cool pack, but avoid placing the vial directly against ice.
Travel planning also includes spare syringes, glucose testing supplies, and a copy of the prescription if required. If daily routines change across time zones, timing adjustments should be reviewed with the prescriber or diabetes care team.
Side Effects and Safety
The most common safety issue with any basal insulin is hypoglycemia (low blood sugar). Symptoms can include shakiness, sweating, hunger, headache, fast heartbeat, blurred vision, confusion, irritability, or unusual tiredness.
Risk can rise with missed meals, extra activity, alcohol use, or changes in other diabetes medicines. Injection-site reactions and weight gain can also occur. Some people notice redness, itching, or mild soreness where the dose was given.
- Common effects: low sugar, site reactions, weight gain
- Serious effects: severe hypoglycemia, allergy, low potassium
- Pattern changes: illness, missed food, dose errors
More serious problems may include severe low blood sugar, allergic reactions, or low potassium. Swelling and heart failure risk can rise when insulin is used with certain thiazolidinedione medicines. Missed doses or handling errors may lead to high blood sugar; persistent vomiting, deep thirst, heavy urination, or worsening illness deserve urgent review. The Acute Hyperglycemia resource covers warning signs in broader terms.
Seek urgent medical care if there is trouble breathing, swelling of the face or throat, fainting, seizure, or confusion that does not improve after suspected low blood sugar is treated. Safety planning is especially important for children, older adults, and anyone with reduced awareness of falling glucose levels.
Drug Interactions and Cautions
Insulin needs can change when other medicines are started or stopped. Corticosteroids, some diuretics, certain antipsychotics, thyroid medicines, and other drugs that raise glucose may increase insulin requirements. Other diabetes medicines can raise the chance of low blood sugar when combined with a long-acting insulin.
Beta blockers can mask some warning signs of hypoglycemia, making episodes harder to recognize. Alcohol can affect glucose levels unpredictably, and kidney or liver impairment can change insulin needs over time. A complete medication list should include prescription drugs, nonprescription products, and supplements.
This vial is for subcutaneous use only. It should not be used in an insulin pump, given intravenously, or relied on for diabetic ketoacidosis treatment unless a clinician directs care in an appropriate medical setting.
Compare With Alternatives
Lantus Vial differs from pen devices mainly in how doses are prepared. A vial and syringe may suit people who prefer manual dose measurement or who already use syringes comfortably. Pens can be simpler for some users, especially when vision, hand strength, or portability is a concern.
Other insulin glargine products may play a similar clinical role but can differ in device design, formulary status, and substitution rules. For some people with type 2 diabetes, clinicians may also use non-insulin medicines alongside or instead of basal insulin, depending on diagnosis and current control.
Examples on this site include Metformin and Jardiance. These are not direct substitutes for everyone, and they do not replace insulin in type 1 diabetes. The broader Diabetes Medications Guide and the Diabetes Medications hub can help when comparing classes, monitoring needs, and common safety differences.
Prescription, Pricing and Access
For many patients, the practical question with Lantus Vial is whether the prescription, quantity, and vial presentation all match the intended treatment plan. Dispensing is handled by licensed partner pharmacies where permitted. Records may need to confirm the product form, dose instructions, and prescriber details before the medicine can move forward.
Coverage varies by plan, formulary status, and whether a payer prefers a specific brand or presentation. Prior authorization or verification steps may apply. For patients paying without insurance, cash-pay options may matter, but the total amount can still change with location, pharmacy, and required documentation.
Stable site-wide updates, when available, may appear on the Promotions Information page. Brand insulin may also have access differences compared with other insulin glargine products. When a prescription is written for a vial, changing to a pen or another insulin is not automatic.
That kind of switch should be reviewed by the prescriber and dispensing pharmacy rather than assumed during refill processing.
Authoritative Sources
Current prescribing details appear in the FDA-approved prescribing information.
Manufacturer handling steps for vial use appear in the vial and syringe injection instructions.
Temperature-sensitive insulin may require prompt, express, cold-chain shipping after dispensing.
This content is for informational purposes only and is not a substitute for professional medical advice.
Express Shipping - from $25.00
Shipping with this method takes 3-5 days
Prices:
- Dry-Packed Products $25.00
- Cold-Packed Products $35.00
Standard Shipping - $15.00
Shipping with this method takes 5-10 days
Prices:
- Dry-Packed Products $15.00
- Not available for Cold-Packed products
Can Lantus come in a vial?
Yes. Lantus is available in a multidose vial presentation as well as pen devices in some settings. This page refers to the 10 mL vial of insulin glargine 100 units/mL. A vial is used with a syringe, so the prescription and the supplied device need to match the intended format. If a prescription was written for a pen instead, the prescriber or dispensing pharmacy may need to confirm whether the vial presentation is appropriate before it is processed.
How many units are in one vial?
A 10 mL vial at 100 units/mL contains 1,000 units in total. That number explains the full amount in the container, but it does not tell how many days it will last for a specific person. The actual duration depends on the prescribed daily dose and the labeled in-use discard period. Once a vial has been opened, the official storage and discard instructions still apply even if insulin remains in the vial.
How long does an opened vial last?
Opened Lantus vials generally follow a 28-day in-use period when stored according to the label. That means a vial may need to be discarded after 28 days even if it is not empty. Unopened vials are typically kept refrigerated until use. Heat, freezing, or storage outside the labeled conditions can affect product integrity, so the carton and official instructions should be checked whenever storage conditions are uncertain.
What side effects need urgent medical attention?
Urgent assessment is important for signs of severe hypoglycemia, such as confusion, fainting, seizure, or symptoms that do not improve after initial treatment for low blood sugar. Trouble breathing, swelling of the face or throat, or widespread rash can suggest a serious allergic reaction. Ongoing vomiting, marked dehydration, or worsening high blood sugar symptoms also deserve prompt medical review. Not every side effect is an emergency, but sudden severe symptoms should not be watched passively at home.
What should be discussed with the prescriber before using the vial?
Useful topics include the exact prescribed dose, whether the prescription is for a vial or a pen, syringe technique, timing of once-daily dosing, and how to handle missed doses according to the official instructions. It is also important to review other diabetes medicines, past severe low blood sugar, kidney or liver problems, pregnancy status if relevant, and whether visual or hand-dexterity issues make a vial and syringe harder to use safely. Those details can affect both safety and product fit.
Can insulin be stopped once blood sugar improves?
That depends on the type of diabetes and the treatment plan. People with type 1 diabetes should not stop basal insulin on their own, because insulin is required for survival. Some people with type 2 diabetes may have treatment changes over time, including less insulin or a shift to other medicines, but that decision depends on diagnosis, glucose patterns, weight, other illnesses, and clinician review. Insulin should never be stopped or reduced without professional guidance.
Rewards Program
Earn points on birthdays, product orders, reviews, friend referrals, and more! Enjoy your medication at unparalleled discounts while reaping rewards for every step you take with us.
You can read more about rewards here.
POINT VALUE
How to earn points
- 1Create an account and start earning.
- 2Earn points every time you shop or perform certain actions.
- 3Redeem points for exclusive discounts.
You Might Also Like
Related Articles
Generic Liraglutide For Weight Loss: A Practical Guide
People searching for generic liraglutide for weight loss usually want clear answers on three points: whether an approved lower-cost version exists, how it differs from brand-name products, and what access…
Liraglutide Vs Semaglutide Comparison Guide For Patients
Key Takeaways People searching liraglutide vs semaglutide usually want a practical comparison, not a slogan. The most useful differences involve formulation, schedule, approved uses, side effect patterns, and the paperwork…
Generic Zepbound: Availability, Status, and Options
Key Takeaways Patients and caregivers often search for generic zepbound when they need a clear answer about availability, labeling, and lower-cost access. The term is common online, but it does…
Oral Wegovy Explained: Facts, Risks, and Access Options
The phrase oral wegovy is best understood as a search term, not a settled product name. Most people use it to mean a pill version of semaglutide for weight management,…