Please note: a valid prescription is required for all prescription medication.
This page helps patients evaluate Tresiba FlexTouch Pens before pursuing a purchase. It explains what this long-acting insulin pen is used for, when prescription review may apply, and the main safety and handling points to check first. It is a product page for people exploring how to buy this basal insulin pen, compare U-100 and U-200 presentations, and understand storage, common risks, and access requirements. Some patients explore US delivery from Canada when eligibility and jurisdiction allow.
How to Buy Tresiba FlexTouch Pens and What to Know First
This product contains insulin degludec, a long-acting basal insulin used to improve blood sugar control in people with diabetes. Before pursuing a purchase, confirm the prescribed concentration, current insulin regimen, and whether other glucose-lowering medicines are also being used. Prescription details may be checked with the prescriber when required.
The FlexTouch device is a prefilled pen injector. The insulin cartridge is built into the pen, and a separate needle is attached for each dose. Because this is a specific device format, the prescription should match both the medicine and the pen presentation, not just the brand name alone.
Product matching matters. FlexTouch pens are available in different concentrations, and confusion between insulin products or strengths can lead to serious dosing errors. Review the carton label, pen label, expiration date, and storage plan before first use. This pen is not used for diabetic ketoacidosis or for rapid correction of severe hyperglycemia.
Who It’s For and Access Requirements
This medicine may be appropriate for adults and for children 1 year and older who need background insulin coverage. In type 1 diabetes it is usually part of a broader insulin plan, while in type 2 diabetes it may be used alone or with other medicines. People reviewing wider Diabetes care, Type 2 Diabetes treatment, or the site’s Diabetes Medications hub may find it easier to compare where basal insulin fits.
Access usually starts with a valid prescription that matches the requested pen concentration and quantity. Extra review may be needed when switching from another insulin, when a recent hospitalization changed the regimen, or when the medical record and prescription do not match. People with visual limitations, reduced hand strength, or caregiver-administered injections may also need device teaching before a refill is pursued.
This product is not intended to replace rapid-acting mealtime insulin in people who need both types. It may also be a poor fit when someone cannot safely read the dose window or use the pen without help. In those cases, caregiver support and device training can matter as much as the prescription itself.
Dosage and Usage
Tresiba FlexTouch Pens are designed for once-daily subcutaneous injections. The number of units is individualized by the prescriber and may differ based on prior insulin use, glucose patterns, age, kidney or liver function, and the rest of the diabetes regimen. This product is a background insulin, so it is not used in the same way as a rapid-acting mealtime insulin.
Each dose should follow the current instructions for use supplied with the pen. The usual steps include checking the label, attaching a new compatible pen needle, priming as directed, dialing the prescribed units, and injecting into the abdomen, thigh, or upper arm with site rotation. The dose window shows actual units. Counting clicks or transferring insulin from the pen into a syringe can cause major dosing mistakes. Needle gauge and length are chosen separately based on the supplied needle product.
Quick tip: Use a new needle for each injection and remove it after the dose is given.
- Check label first – confirm the prescribed concentration
- Prime as directed – confirm insulin flow
- Rotate sites – lower skin thickening risk
- Read the dose window – use the displayed units
Strengths and Forms
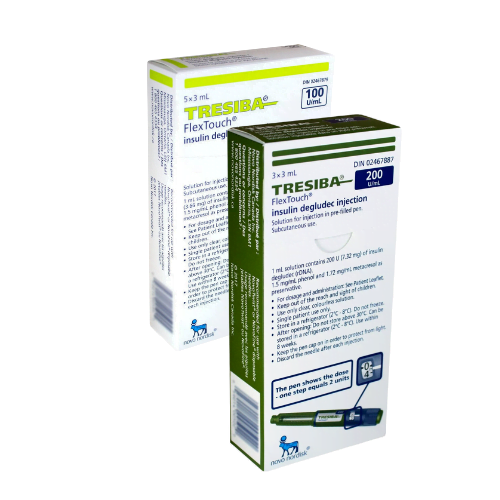
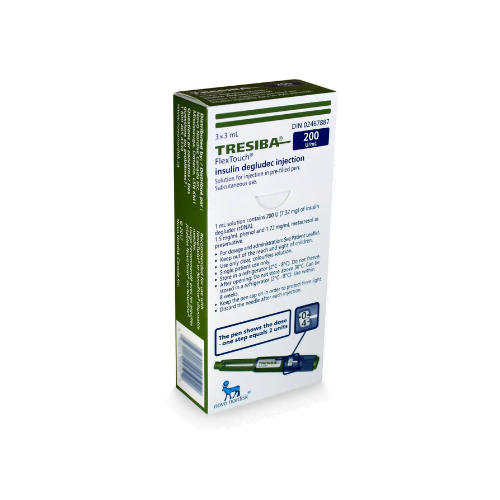
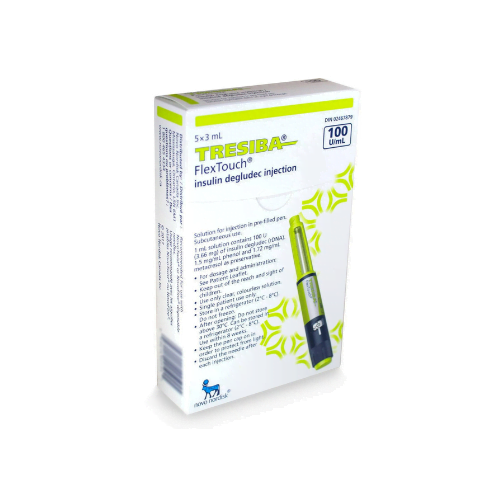
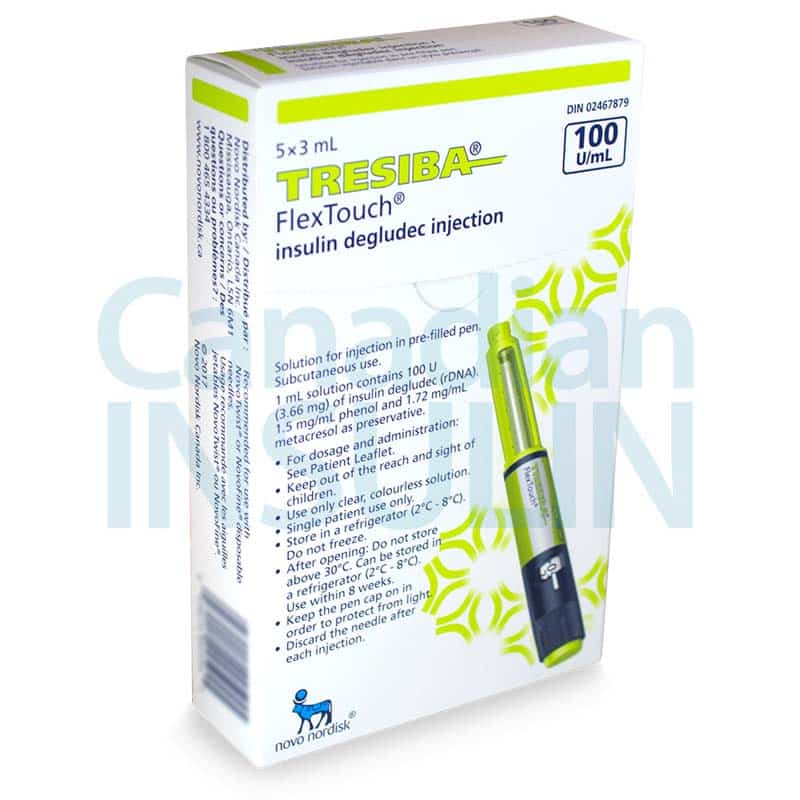
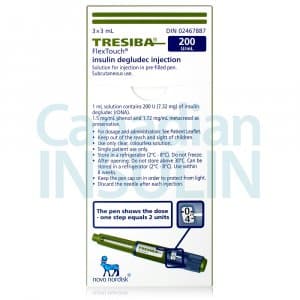
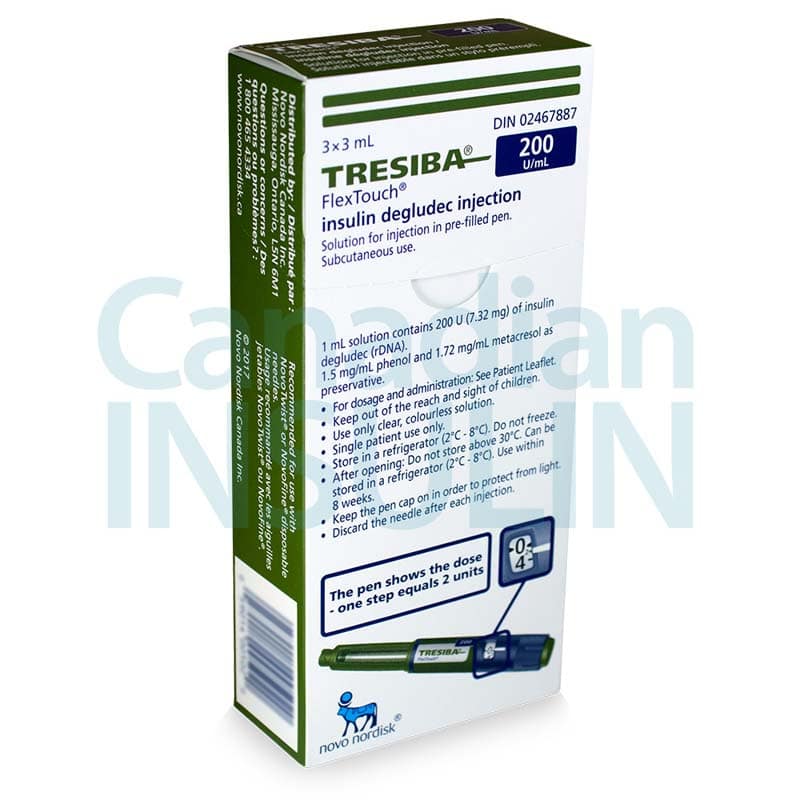
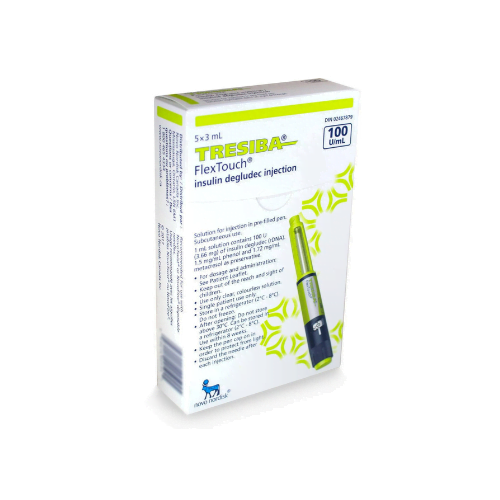
Tresiba FlexTouch Pens are commonly supplied as 3 mL prefilled insulin pens in two concentrations: U-100 and U-200. The concentration changes how much insulin is contained in each mL, so the carton and pen label must match the prescription. Availability can vary by pharmacy and jurisdiction, especially when a specific concentration is requested.
Each 3 mL U-100 pen contains 300 units of insulin degludec, while each 3 mL U-200 pen contains 600 units. Cartons are commonly packaged as five pens, but the dispensed quantity can still vary with the prescription and local supply practices. For many patients, the practical question is whether the prescribed number of units, refill amount, and concentration are correct. The pen display shows actual units, so separate dose conversion should not be attempted unless the prescriber changes products.
FlexTouch is a complete prefilled pen rather than a cartridge that fits into a separate reusable pen body. That difference matters for people comparing cartridge systems, because the device handling, replacement parts, and prescription wording are not the same.
| Option | Insulin per pen | Presentation | Practical note |
|---|---|---|---|
| U-100 | 300 units | 3 mL prefilled pen | Used when standard unit increments are prescribed |
| U-200 | 600 units | 3 mL prefilled pen | More concentrated for higher daily unit requirements |
Storage and Travel Basics
Insulin handling is part of safe use. Unopened pens are generally stored in the refrigerator and should never be frozen. A pen that has been frozen, overheated, or physically damaged should not be used. Keep the cap on when the pen is not in use and protect it from direct heat and light.
Once a pen is opened, follow the current carton and instructions for room-temperature or refrigerated storage. Many current labels allow an in-use pen for up to 56 days when stored as directed, but the package insert should still be checked because storage language can change. Writing the first-use date on the carton can help avoid accidental overuse.
For trips, insulin is usually safer in hand luggage than in checked baggage when that option is available. Keeping supplies in their original packaging can make label checks easier. A backup pen, extra needles, and glucose monitoring supplies can also reduce interruptions if travel runs longer than expected.
- Unopened pens – refrigerate and do not freeze
- In-use pens – store only as the label directs
- Travel packing – keep with prescription labeling
- Heat exposure – avoid cars, windows, and direct sun
Side Effects and Safety
The main safety concern with any insulin is hypoglycemia, or low blood sugar. Symptoms can include sweating, shaking, hunger, headache, dizziness, confusion, irritability, or blurred vision. The risk may rise after dose changes, reduced food intake, unusual exercise, alcohol use, or when insulin is combined with other glucose-lowering medicines. Glucose monitoring should follow the prescriber’s plan.
Other side effects may include injection-site reactions, swelling, weight change, mild cold-like symptoms, or headache. More serious problems can include severe hypoglycemia, allergic reactions, and low potassium. Repeated injections into the same spot can also lead to skin changes that affect absorption. Anyone with trouble breathing, widespread rash, facial swelling, fainting, or seizures needs urgent medical care.
Persistent unexpected low or high readings, especially after switching products or concentrations, deserve clinical review. Keeping the pen label and current medication list together can help identify whether the issue involves the dose, timing, storage, or a mix-up with another insulin.
Why it matters: Pen mix-ups and skipped monitoring can turn a routine refill into a safety problem.
- Common issues – low blood sugar, local irritation, headache
- Serious signs – severe weakness, confusion, trouble breathing
- Pen safety – never share the device with others
Drug Interactions and Cautions
Insulin needs can change when other medicines are added or stopped. Drugs that may affect glucose include oral diabetes medicines, GLP-1 therapies, SGLT2 inhibitors, corticosteroids, diuretics, and some blood pressure medicines. Beta blockers can sometimes hide warning signs of low blood sugar, which makes regular monitoring more important. Alcohol can also make glucose readings less predictable.
People using combination therapy can review broader class context in Common Diabetes Medications, Diabetes Medications List, and SGLT2 Inhibitors. Extra caution is also needed with recurrent hypoglycemia, kidney or liver impairment, acute illness, or reduced food intake.
A current medication list is useful whenever a new medicine is added, stopped, or adjusted. That includes non-diabetes medicines and supplements, since illness treatment, steroids, and some OTC products can change glucose patterns or appetite. Thiazolidinedione medicines used with insulin may also increase fluid retention risk in some patients.
- Combination therapy – may change glucose patterns
- Beta blockers – can mask low sugar symptoms
- Fluid retention risk – watch insulin plus TZD combinations
Compare With Alternatives
Basal insulin is not a direct substitute for every non-insulin treatment. Metformin lowers glucose through a different pathway and is often used earlier in type 2 diabetes. Farxiga Dapagliflozin is an SGLT2 inhibitor that works through the kidneys and may be chosen for some patients with broader metabolic or organ-related treatment priorities. These are different treatment classes, so changes should be clinician-led rather than self-directed.
When clinicians compare basal insulin options, they usually focus on concentration, dose flexibility, pen design, prior hypoglycemia history, and how the rest of the regimen is built. In type 1 diabetes, basal insulin still needs to fit with mealtime insulin. In type 2 diabetes, the decision may involve whether insulin is being added to oral therapy or replacing another long-acting insulin.
Within the insulin category, comparisons are often about device preference, daily dose size, and how flexible the dosing schedule needs to be, not just brand name alone. A prescriber may also weigh prior reactions, formulary rules, and whether a concentrated pen would simplify the regimen.
Prescription, Pricing and Access
When comparing Tresiba FlexTouch Pens, check that the prescription lists the correct concentration, quantity, and directions. This site works as a prescription referral service rather than an in-house dispensing pharmacy. Licensed third-party pharmacies handle dispensing where permitted. A mismatch between U-100 and U-200, or between pens and vials, can delay review and create safety concerns.
For patients reviewing Tresiba FlexTouch cost without insurance, the final amount can vary by pharmacy, concentration, quantity, and local requirements. Some people also compare cash-pay arrangements when coverage is limited. Documentation such as the prescription, patient details, and current medication list may be needed so the dispensed pen matches the prescriber’s order.
Pen needles may be supplied separately, so it helps to confirm whether they are included in the prescription plan. Concentration checks, patient verification, and jurisdiction rules can all affect whether a request can proceed, even when the brand name is clear.
General program details may appear on the Promotions Information page, but terms can change and availability is not guaranteed. Cross-border arrangements may depend on eligibility and jurisdiction.
Authoritative Sources
- Official patient device instructions are available here: Tresiba FlexTouch pen instructions
- Prescribing and administration details can be reviewed here: NovoMedLink Tresiba FlexTouch and vial details
- Independent diabetes device reference can be reviewed here: ADCES pen detail
Where dispensing is permitted, temperature-sensitive insulin may require prompt, express, cold-chain shipping arranged by the dispensing pharmacy.
This content is for informational purposes only and is not a substitute for professional medical advice.
Express Shipping - from $25.00
Shipping with this method takes 3-5 days
Prices:
- Dry-Packed Products $25.00
- Cold-Packed Products $35.00
Standard Shipping - $15.00
Shipping with this method takes 5-10 days
Prices:
- Dry-Packed Products $15.00
- Not available for Cold-Packed products
How many pens are usually in a box of Tresiba FlexTouch?
In many markets, Tresiba FlexTouch is commonly packaged as a carton of five prefilled 3 mL pens. The final quantity dispensed can still vary based on the prescription, local packaging rules, and pharmacy stock. The safest way to confirm is to read the carton label and prescription directions together. It is also important to verify whether the box contains U-100 or U-200 pens, since those presentations are different and should only be used as prescribed.
How long can a Tresiba FlexTouch pen be used after first use?
Current labeling generally allows an in-use Tresiba FlexTouch pen for up to 56 days when stored as directed, but the exact storage instructions on the carton and package insert should always be checked. Storage rules may differ for unopened and in-use pens. Writing down the first-use date can help avoid accidental use beyond the labeled period. A pen that has been frozen, overheated, damaged, or kept past its expiration date should not be used.
What should be avoided while using Tresiba?
Avoid sharing the pen with anyone else, even if the needle is changed. It is also important to avoid using the wrong concentration, using insulin that has been frozen or overheated, or changing the dose without clinical guidance. Alcohol can make blood sugar less predictable, and some medicines can change insulin needs or mask low blood sugar symptoms. Careful label checks and routine glucose monitoring are key parts of safe use.
What should be discussed with a clinician before starting this pen?
Important topics include the current insulin regimen, recent low blood sugar episodes, kidney or liver problems, pregnancy, major illness, and any recent medication changes. It also helps to ask about the right concentration, when to monitor glucose, whether mealtime insulin is also needed, and how to store the pen once opened. If someone has vision, dexterity, or caregiver-support issues, device teaching should be part of the conversation as well.
What needles work with a Tresiba FlexTouch pen?
Tresiba FlexTouch is used with compatible insulin pen needles, which are attached separately for each injection. The exact needle gauge and length can vary based on the supplied needle product, individual preference, and clinical guidance. It is best to confirm needle compatibility in the current instructions for use and on the pharmacy label. A new needle should be used for every injection, and the needle should be removed after the dose is given.
What kind of pen is used for Tresiba insulin?
For this presentation, Tresiba comes as a FlexTouch prefilled pen injector that contains insulin degludec inside the device. That means the insulin cartridge is built into the pen, rather than placed into a separate reusable pen body. References may also discuss vial presentations, but this page is about the prefilled pen format. The exact presentation matters because prescription wording, device handling, and concentration checks should match the product that is actually dispensed.
Rewards Program
Earn points on birthdays, product orders, reviews, friend referrals, and more! Enjoy your medication at unparalleled discounts while reaping rewards for every step you take with us.
You can read more about rewards here.
POINT VALUE
How to earn points
- 1Create an account and start earning.
- 2Earn points every time you shop or perform certain actions.
- 3Redeem points for exclusive discounts.
You Might Also Like
Related Articles
Generic Liraglutide For Weight Loss: A Practical Guide
People searching for generic liraglutide for weight loss usually want clear answers on three points: whether an approved lower-cost version exists, how it differs from brand-name products, and what access…
Liraglutide Vs Semaglutide Comparison Guide For Patients
Key Takeaways People searching liraglutide vs semaglutide usually want a practical comparison, not a slogan. The most useful differences involve formulation, schedule, approved uses, side effect patterns, and the paperwork…
Generic Zepbound: Availability, Status, and Options
Key Takeaways Patients and caregivers often search for generic zepbound when they need a clear answer about availability, labeling, and lower-cost access. The term is common online, but it does…
Oral Wegovy Explained: Facts, Risks, and Access Options
The phrase oral wegovy is best understood as a search term, not a settled product name. Most people use it to mean a pill version of semaglutide for weight management,…