Please note: a valid prescription is required for all prescription medication.
Buy Beovu Pre-filled Syringe online with a valid prescription and compare current listed pricing, presentation details, and safety basics before ordering. You can use this page to check the brolucizumab prefilled syringe format, match the selected option to your prescription, and review handling notes for an injectable eye medicine. If you are comparing access with US delivery from Canada, keep the prescriber and order details available before checkout.
Beovu Pre-filled Syringe Price and Available Options
The Beovu price shown on the product listing should be compared with the exact presentation selected at checkout. For a specialty eye injection, the important details are the form, strength, concentration, quantity, and whether the listing is for a prefilled syringe rather than a vial or another anti-VEGF medicine. Current listed pricing may change with the selected product option and order details, so match the product label to the wording on your clinician’s order.
Beovu is commonly identified as a 6 mg/0.05 mL intravitreal injection with a 120 mg/mL concentration. Some product information also describes the total syringe contents, because a prefilled syringe can contain more solution than the delivered intravitreal dose. That distinction matters when comparing Beovu injection price across listings: the concentration, total fill volume, and usable delivered amount are not the same thing.
If you are comparing Beovu cost without insurance, focus on the current product amount, quantity, shipping or handling charges, and any coverage paperwork requested during checkout. Cash-pay comparisons should still start with the prescribed presentation, not only the lowest displayed number. A different retinal medicine, vial format, or strength listing may not be interchangeable with a brolucizumab injection.
| Product detail | What to check |
|---|---|
| Active ingredient | Brolucizumab, an anti-VEGF medicine for selected retinal conditions. |
| Common presentation | Single-dose prefilled syringe for intravitreal use by an eye care professional. |
| Strength wording | Often listed as 6 mg/0.05 mL or 120 mg/mL. |
| Selection step | Match the form, strength, and quantity to the prescription before checkout. |
| Practical note | Total syringe contents may differ from the delivered dose used in the eye. |
Quick tip: Compare the selected presentation and quantity before comparing the final product amount.
How to Buy Beovu Online
When you buy Beovu online, start with the product form. This item is a prefilled syringe, not an eye drop, tablet, or self-injection device for home use. The selected listing should match the medicine name, strength, and presentation written by the eye specialist. If the order wording includes brolucizumab-dbll, brolucizumab, or Beovu 6 mg pre-filled syringe, confirm that the listed product aligns with the exact wording before continuing.
This prescription item cannot be completed without appropriate prescriber information. Prescriber details may be confirmed when needed for a prescription order, and supporting documents may be requested for some orders. Keep clinic contact information available so any clarification can be handled without changing the product selection yourself.
- Choose the form: Select the prefilled syringe only when it matches the order.
- Check the strength: Confirm 6 mg/0.05 mL or other listed strength wording.
- Review quantity: Make sure the number selected matches the intended supply.
- Prepare clinic details: Keep prescriber contact information ready if confirmation is needed.
- Read handling notes: Specialty injectables may need temperature-controlled storage.
Do not substitute another anti-VEGF medicine based only on convenience or cost. Retina clinics may use different products for different clinical reasons, and the injected medicine must match the treatment plan.
Product Details to Match Before Checkout
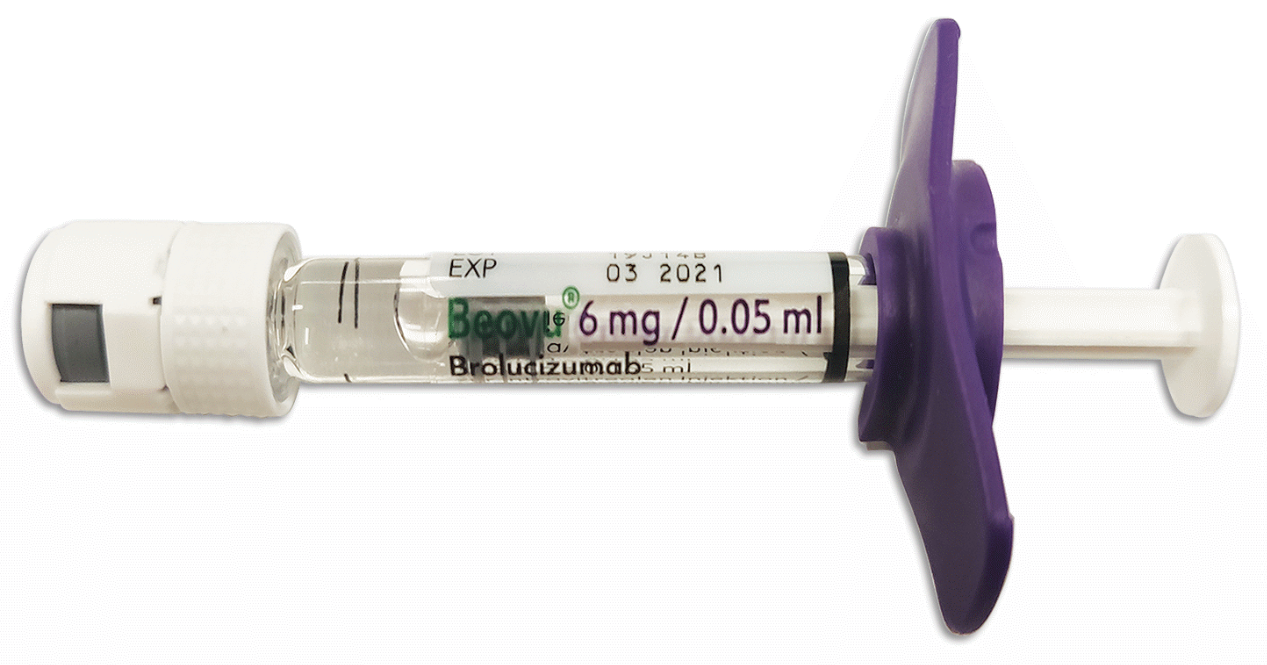
Beovu is a sterile, single-dose eye injection supplied for intravitreal administration, which means injection into the vitreous gel inside the eye. It is prepared and administered by a trained eye care professional under sterile conditions. The prefilled syringe format can help clinics avoid some preparation steps, but it does not make the medicine suitable for patient self-injection.
The Beovu 6 mg/0.05 mL pre-filled syringe is associated with a 120 mg/mL concentration. The syringe is intended for single use, and unused solution is discarded according to clinical handling procedures. If your clinic’s order names a vial, a different anti-VEGF agent, or a different strength, do not assume this prefilled option is the right listing.
Product labels may use both brand and generic names. Beovu is the brand name, while brolucizumab is the active ingredient. In some United States prescribing materials, the ingredient may be written as brolucizumab-dbll. These names are closely related, but the presentation and strength still need to match the selected product.
Why it matters: Matching the exact presentation reduces avoidable delays and product-selection errors.
What This Eye Injection Is Used For
Beovu is an anti-VEGF injection. Anti-VEGF medicines block vascular endothelial growth factor, a signal that can contribute to abnormal blood vessel growth and leakage in the retina. The medicine is used by retina specialists for neovascular, or wet, age-related macular degeneration and diabetic macular edema when clinically appropriate.
Customers comparing Beovu for wet AMD can also review the site’s Wet Age Related Macular Degeneration product list. Those comparing Beovu for diabetic macular edema can browse Diabetic Macular Edema options. These pages are useful for navigation, but the final treatment choice should come from the prescribing eye specialist.
This medicine is not a general treatment for every type of blurry vision, eye pain, or retinal disease. A diagnosis, retinal imaging, and treatment history often guide whether a clinician selects Beovu, another anti-VEGF injection, laser treatment, observation, or a different plan. Use the product page to match the prescribed product, not to decide whether treatment is needed.
Storage, Handling, and Shipping Basics
Beovu prefilled syringes require careful handling because they are sterile injectable biologic products. Product information commonly directs storage in a refrigerator at 2°C to 8°C, or 36°F to 46°F, with protection from light. The syringe should not be frozen, shaken, or used if the packaging appears damaged. Your clinic or dispensing instructions should guide final handling before administration.
Plan the order around the clinic’s appointment schedule, but avoid assuming a treatment date until the product details and access steps are complete. A retina clinic may need time to inspect the syringe, prepare the injection setting, and follow its own sterile-use procedures. If a package arrives with special storage instructions, follow them promptly and contact the appropriate care team if temperature control seems compromised.
Shipping options may include express cold-chain handling when temperature control is required. This is a logistics consideration, not a promise about arrival time or product availability. For specialty eye medicines, the most practical customer step is to verify the selected quantity, storage expectations, and clinic handoff before the order is finalized.
- Temperature: Follow refrigerated storage instructions when supplied.
- Light protection: Keep the product in protective packaging until use.
- Single use: The syringe is not intended for repeated use.
- Clinic handling: Administration should occur in a sterile clinical setting.
- Travel planning: Ask the clinic how to manage timing and transport.
Safety Checks Before Ordering
Review safety basics before ordering any intravitreal injection. Beovu should not be used in people with ocular or periocular infections, active inflammation inside the eye, or known hypersensitivity to brolucizumab or product components. These are important checks because the medicine is injected directly into the eye by a clinician.
Serious eye-related risks have been reported with intravitreal anti-VEGF therapy. These can include endophthalmitis, a severe eye infection; retinal detachment; increased pressure inside the eye; retinal vasculitis, which is inflammation of retinal blood vessels; and retinal vascular occlusion, which is blockage of blood flow in retinal vessels. The official prescribing information also discusses arterial thromboembolic events, such as stroke-related or heart-related vascular events, as a class concern.
More common post-injection effects can include eye discomfort, blurred vision, floaters, conjunctival bleeding, or temporary changes in vision. Some symptoms are expected to be checked after an eye injection, while others may signal an urgent problem. Seek prompt medical attention if severe eye pain, worsening redness, light sensitivity, a sudden drop in vision, new flashes, a curtain-like shadow, or a marked increase in floaters occurs.
Tell the eye specialist about recent eye surgery, current eye infection symptoms, prior inflammatory reactions after injections, stroke or heart attack history, pregnancy, breastfeeding, or any planned procedures. Also provide a current medication list, including blood thinners and over-the-counter products. This information helps the clinic decide whether treatment timing, monitoring, or an alternative plan should be considered.
Monitoring and Follow-Up Questions
Beovu dosing and treatment intervals are determined by the retina specialist. The label includes scheduled intravitreal dosing, but individual plans may differ based on diagnosis, imaging results, response, and safety findings. Do not change injection timing, skip follow-up visits, or extend intervals based on product-page information.
Monitoring usually focuses on vision changes, retinal fluid, eye pressure, inflammation, and vascular events. If diabetes is part of the reason for eye treatment, it may help to understand how blood sugar and retinal disease connect through How Does Diabetes Affect The Eyes. People tracking retinal symptoms can also use Diabetic Retinopathy Signs Symptoms as a practical symptom-awareness resource.
Before the next appointment, ask which symptoms should trigger an urgent call, how vision will be monitored, and whether the same anti-VEGF medicine is expected to continue. These questions are more useful than comparing dose schedules alone, because retinal treatment plans often depend on imaging and safety response.
Comparing Anti-VEGF Options
Searches such as Beovu vs Eylea or Beovu vs Vabysmo usually reflect a real product decision, but the comparison is not only about strength or price. These medicines use different active ingredients, dosing schedules, label details, and safety profiles. A retina specialist may choose one option over another based on diagnosis, prior response, inflammation risk, convenience, or clinic protocol.
For product browsing, the Ophthalmology category groups eye-related products available on the site. Use it to compare prescribed ophthalmic options, not to replace a clinician’s recommendation. If another product name appears on your order, select that specific listing instead of substituting Beovu on your own.
When comparing Beovu cost with other retinal medicines, also compare the prescribed presentation, number of syringes or vials, storage needs, and clinic administration plan. A medicine with a similar treatment category may still be inappropriate if the diagnosis, safety history, or prescriber order is different.
Authoritative Sources
Official product information can help you confirm label-level details before discussing treatment with the eye clinic. Official Beovu prescribing information summarizes indications, administration, contraindications, warnings, and storage requirements.
UK product information provides additional wording on the prefilled syringe presentation and supplied concentration. Beovu summary of product characteristics describes the 120 mg/mL solution for injection in a prefilled syringe.
Use these sources to support questions for a licensed clinician, especially if the product label, diagnosis, or syringe presentation differs from the order details in front of you.
This content is for informational purposes only and is not a substitute for professional medical advice.
{acf_product_technical_information}
Express Shipping - from $25.00
Shipping with this method takes 3-5 days
Prices:
- Dry-Packed Products $25.00
- Cold-Packed Products $35.00
Standard Shipping - $15.00
Shipping with this method takes 5-10 days
Prices:
- Dry-Packed Products $15.00
- Not available for Cold-Packed products
What is Beovu injection used for?
Beovu is an intravitreal anti-VEGF injection used by retina specialists for certain retinal diseases, including wet age-related macular degeneration and diabetic macular edema. Intravitreal means the medicine is injected into the gel-like space inside the eye. It is not an eye drop or a self-administered medicine. A clinician usually confirms the diagnosis with eye examination and imaging before deciding whether Beovu or another treatment is appropriate.
Is Beovu still on the market?
Beovu remains an available prescription retinal medicine in some markets and presentations, including prefilled syringe formats. Availability can differ by country, supplier, product presentation, and clinical use. The medicine also carries important safety warnings, including risks of retinal vasculitis and retinal vascular occlusion. Anyone considering treatment should rely on current prescribing information and the treating retina specialist’s assessment rather than older product announcements or general web summaries.
Is the Beovu pre-filled syringe single use only?
Yes. The Beovu pre-filled syringe is intended as a sterile, single-dose presentation for intravitreal administration by an eye care professional. It may contain more solution than the amount delivered into the eye, so total fill volume should not be confused with the injected dose. Any unused portion is handled according to clinical procedures. Patients should not try to prepare, adjust, or inject the syringe themselves.
What symptoms after a Beovu injection need urgent attention?
Urgent symptoms can include severe eye pain, worsening redness, light sensitivity, sudden vision loss, new flashes, a curtain-like shadow, or a major increase in floaters. These symptoms may suggest infection, retinal detachment, inflammation, or a vascular problem and should be assessed promptly. Some temporary discomfort or blurred vision can occur after eye injections, but a clinician should explain which symptoms are expected and which require immediate contact.
What should I ask my eye specialist before starting Beovu?
Useful questions include why Beovu is being selected, how response will be monitored, what symptoms should trigger urgent contact, and whether prior inflammation or vascular history changes the risk discussion. Ask how the clinic schedules follow-up imaging and whether the prefilled syringe format is the intended presentation. Also mention pregnancy, breastfeeding, recent eye procedures, eye infection symptoms, allergies, and all current medicines, including over-the-counter products.
How is Beovu different from Eylea or Vabysmo?
Beovu, Eylea, and Vabysmo are injectable retinal medicines, but they contain different active ingredients and have different label details, dosing schedules, and safety considerations. A retina specialist may choose among them based on diagnosis, previous response, retinal imaging, inflammation risk, clinic experience, and patient-specific factors. They should not be treated as automatically interchangeable, even when they are discussed within the same anti-VEGF treatment category.
Rewards Program
Earn points on birthdays, product orders, reviews, friend referrals, and more! Enjoy your medication at unparalleled discounts while reaping rewards for every step you take with us.
You can read more about rewards here.
POINT VALUE
How to earn points
- 1Create an account and start earning.
- 2Earn points every time you shop or perform certain actions.
- 3Redeem points for exclusive discounts.
You Might Also Like
Related Articles
Continuous Glucose Monitoring: How CGMs Fit Diabetes Care
Continuous glucose monitoring is a way to track glucose throughout the day and night with a small wearable sensor. It matters because it shows patterns, direction, and alerts that a…
How to Get Rid of a Cold Sore and Ease Symptoms Safely
A cold sore usually cannot be erased overnight. If you want to know how to get rid of a cold sore, the fastest practical step is to treat it early,…
What Is Glucagon Like Peptide 1 and What Does It Do?
What is glucagon like peptide 1? In simple terms, it is a hormone your gut releases after you eat. Clinically, it is called glucagon-like peptide-1, or GLP-1, an incretin (a…
Does Metformin Cause Weight Loss? Expectations and Limits
Yes, metformin can cause modest weight loss in some people, but it is not primarily a weight-loss drug. If you are asking does metformin cause weight loss, the practical answer…